ETH Zürich published a scientific publication about SFERA-III in the international journal PCCP (Physical Chemistry Chemical Physics). The paper features in the January 2020 Issue (4) and is available in open access.
Abstract
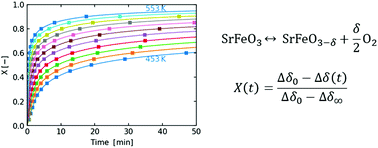
The perovskite oxide SrFeO3 has favourable redox properties for oxygen exchange applications, including oxygen separation and oxygen production chemical looping cycles. For such applications, lower temperature operation can improve the energy demand and feasibility of the process, but can also lead to kinetic limitations. Here we investigate the oxidation and reduction reaction kinetics of SrFeO3 in the temperature range 450–750 K. Isothermal relaxation techniques are used to observe the reaction rates across this temperature range, using a thermogravimetric analysis system.
Experimental data are analysed according to an isoconversional method and fit with a simple power law model to extract activation energies. The apparent activation energy of oxidation and reduction was found to be 92 ± 16 and 144 ± 17 kJ mol−1 respectively. Comparison of oxidation and reduction kinetics together with considerations of particle size indicate that the oxidation reaction rate may be limited by diffusion in the bulk, while the reduction reaction rate is limited by the surface reaction. Furthermore, we also investigated the mixed perovskite Sr0.93Ca0.07Fe0.9Co0.1O3, which exhibited a 4-fold increase in the oxidation rate.
References
Isothermal relaxation kinetics for the reduction and oxidation of SrFeO 3 based perovskites
Author(s): B. Bulfin, J. Vieten, S. Richter, J. M. Naik, G. R. Patzke, M. Roeb, C. Sattler, A. Steinfeld
Published in: Physical Chemistry Chemical Physics, Issue 22/4, 2020, Page(s) 2466-2474, ISSN 1463-9076

This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 823802
